Compare drug prices at GoodRx.ca
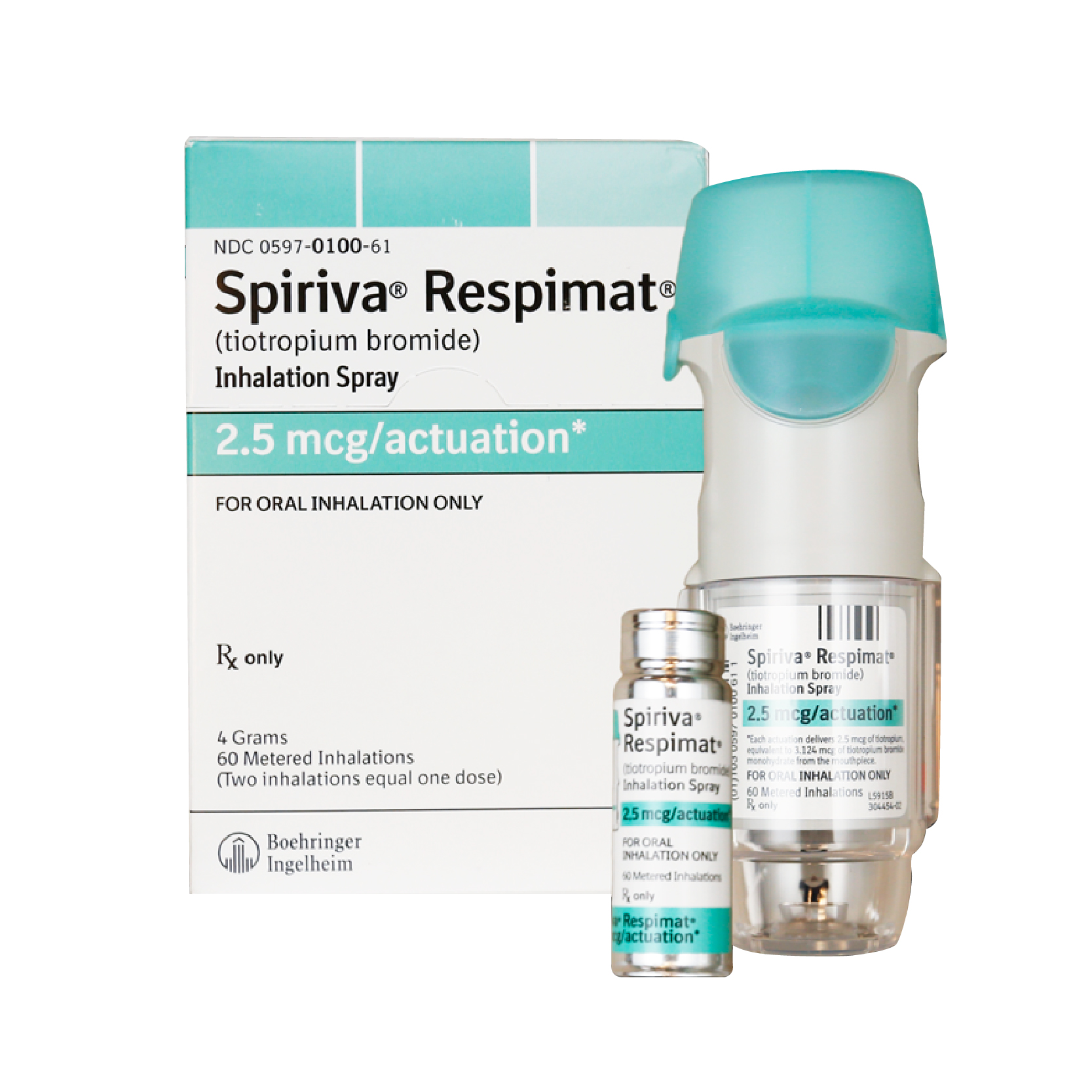
Spiriva Respimat (Tiotropium Bromide) Inhaler 2.5mcg – 60 dose
US121.00
60 Dose
Brand Manufacturer: BOEHRINGER INGELHEIM LTD.
Please click the currency converter flag in the top corner of the screen for the exchange rate from USD-CAD.
Compare CAD-USA drug prices at GoodRx.com
This order requires you to submit a prescription. Upload here. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States. | Fulfillment policy See More.Tiotropium, Spiriva
Description: Bronchodilator, Long-Acting Anticholinergic Agent
CSA NA – FDA Approved – REMS (N) – Can Ship
How Does It Work
Tiotropium/Spiriva inhibits acetylcholine at muscarinic type 3 (M3) receptors in the smooth muscle of the bronchioles resulting in bronchodilation.
Indications For Use
Tiotropium/Spiriva is officially indicated for chronic obstructive pulmonary disease and the Spiriva Respimat device is also indicated for asthma in patients 6 years or older (may also be used for conditions not listed)
Precautions
Before starting Tiotropium/Spiriva make sure your physician is aware of any allergies or medications you currently take, if you have kidney disease, narrow-angle glaucoma, prostate hyperplasia, are pregnant or breastfeeding.
Usual Dosing
Spiriva Handihaler- Inhale content of one capsule (18mcg) once daily, using handihaler device
Spiriva Respimat – Inhale 2 actuations (=5mcg) once daily
Pharmacist Tips On Using
How to Take: Handihaler: Capsule should be placed in handihaler and then the mouthpiece and dustcover should be closed. Press the button on the side to pierce a hole in the capsule, open the dust cover and inhale a long, slow, deep inhalation through the mouthpiece and hold for a few seconds. Repeat a second time to ensure all medication is released.
Respimat: The cartridge should be inserted then the device should be primed by twisting it 180 degrees then pressing the button. Repeat until an aerosol cloud is visible. To take dosage load and press button and inhale the aerosol with a slow, deep breath and hold for a few seconds.
You Need to Avoid: Do not exceed recommended dosages as it may cause severe adverse reactions; do not get in the eyes. Do not breathe out into handihaler device and keep it dry.
Side Effects
Dry mouth, upper respiratory infection, pharyngitis, sinusitis. It may rarely cause paradoxical bronchospasm which may be misinterpreted as lack of effect. If symptoms worsen after use, discontinue the medication.
Note this is not a complete list of side effects, only common ones
Special note for customers.
*All orders are limited to a 90 day supply. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States.Disclaimer
All information contained in and provided by ZIPRx.ca is provided for educational purposes only. This information should not be used for the diagnosis or treatment of any health problem or disease. This information is not intended to replace clinical judgement or guide individual patient care in any manner. The User is hereby notified that the information contained herein may not meet the user’s needs. The User is advised that, although the information is derived from medical research and literature, we cannot guarantee either its correctness, comprehensiveness or currency. ZIPRx nor any of its affiliated parties shall be liable for any damage resulting in whole or part from any User’s use of or reliance upon the information contained within.Shipping Details
All orders received prior to 3:00 pm PST on business days will ship the following day; peak season and holiday shipping days may require an additional 24 hours. Our Pharmacy will make every effort to ship each order as quickly as possible. Orders received over the weekend will ship the following Monday as our shipping department is closed Saturday and Sunday. We offer shipping via Canada Post within Canada. We offer shipping via USPS and UPS within the continental United States and worldwide. Expedited shipping options are available at checkout if you want to receive your order sooner.Related products
Brand Name
Respiratory