Compare drug prices at GoodRx.ca
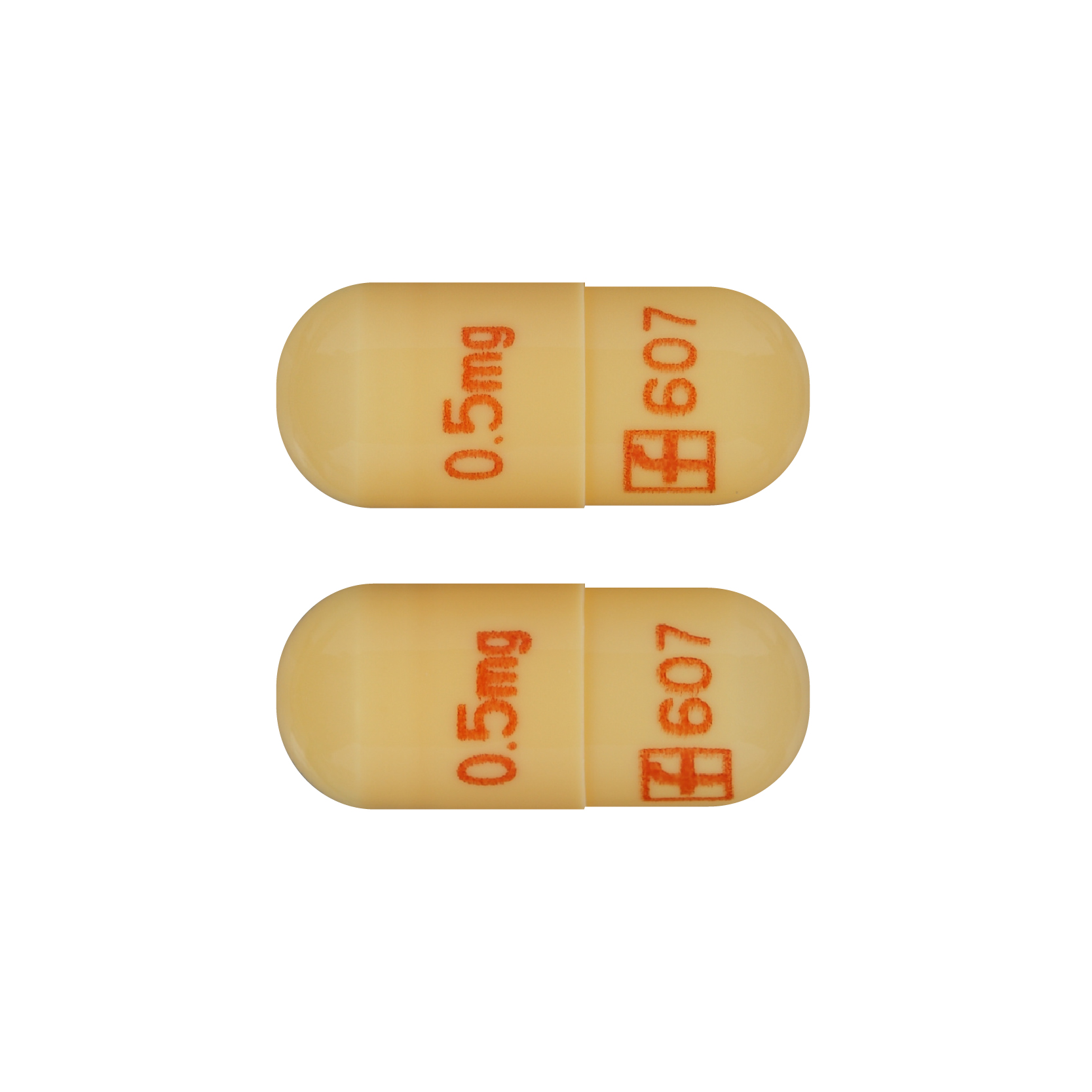
Prograf (Tacrolimus) 0.5mg – 100 cap
US338.00
100 Cap
Brand Manufacturer: ASTELLAS PHARMA CANADA
Please click the currency converter flag in the top corner of the screen for the exchange rate from USD-CAD.
Compare CAD-USA drug prices at GoodRx.com
This order requires you to submit a prescription. Upload here. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States. | Fulfillment policy See More.Calcineurin Inhibitor
“ALERT: US Boxed Warning
Malignancies and serious infection:
Increased risk for developing serious infections and malignancies with tacrolimus or other immunosuppressants that may lead to hospitalization or death.
Mortality in liver transplantation (Astagraf XL):
Increased mortality in female transplant patients with Astagraf XL. Astagraf XL is not approved for use in liver transplantation.”
CSA NA – FDA Approved – REMS (N) – Can Ship
How Does It Work
Tacrolimus suppresses cellular immunity (inhibits T-lymphocyte activation), by binding to an intracellular protein, FKBP-12, and complexes with calcineurin dependent proteins to inhibit calcineurin phosphatase activity. Inhibition of calcineurin also is important in maintaining neuronal and synaptic integrity and homeostasis for peripheral nerve regeneration.
Indications For Use
Officially indicated for organ rejection prophylaxis. It is also prescribed off-label for use in Crohn disease (perianal/fistulizing disease), graft-versus host disease (prevention/treatment), immunosuppression (maintenance) after lung transplant, intestinal transplant, myasthenia gravis, rheumatoid arthritis (refractory), and peripheral nerve regeneration.
Precautions
Do not use in patients with a known hypersensitivity to tacrolimus.
Do not use in patients with congenital long QT syndrome
Use with caution in patients with heart failure, brady arrhythmia, taking antiarrhythmic mediations or other drugs that can lead to QT prolongation
Immunosuppressant agents including tacrolimus can increase the risk of infection leading to hospitalization or death
Immunosuppressive agents including tacrolimus may be associated with the development of malignancies that can lead to hospitalization or death
Usual Dosing
Immunosuppressants such as tacrolimus have a wide range of dosing options. For a comprehensive listing, please check an official drug monograph.
Side Effects
Immunosuppressants such as tacrolimus have an extensive side effect profile. For a comprehensive listing, please check an official drug monograph.
Special note for customers.
*All orders are limited to a 90 day supply. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States.Disclaimer
All information contained in and provided by ZIPRx.ca is provided for educational purposes only. This information should not be used for the diagnosis or treatment of any health problem or disease. This information is not intended to replace clinical judgement or guide individual patient care in any manner. The User is hereby notified that the information contained herein may not meet the user’s needs. The User is advised that, although the information is derived from medical research and literature, we cannot guarantee either its correctness, comprehensiveness or currency. ZIPRx nor any of its affiliated parties shall be liable for any damage resulting in whole or part from any User’s use of or reliance upon the information contained within.Shipping Details
All orders received prior to 3:00 pm PST on business days will ship the following day; peak season and holiday shipping days may require an additional 24 hours. Our Pharmacy will make every effort to ship each order as quickly as possible. Orders received over the weekend will ship the following Monday as our shipping department is closed Saturday and Sunday. We offer shipping via Canada Post within Canada. We offer shipping via USPS and UPS within the continental United States and worldwide. Expedited shipping options are available at checkout if you want to receive your order sooner.Related products
Brand Name
Allergies
Brand Name