Compare drug prices at GoodRx.ca
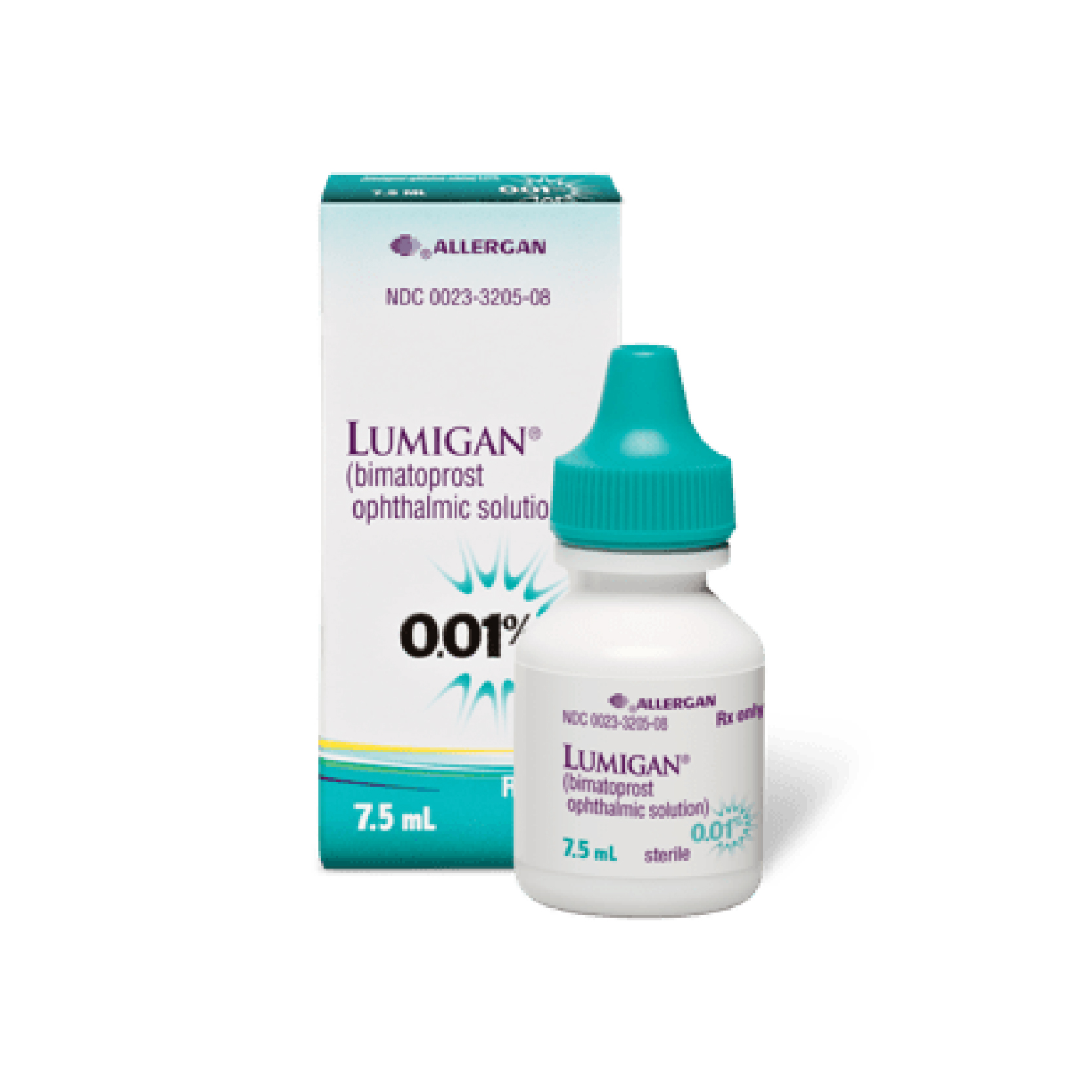
Lumigan RC (Bimatoprost) 0.01% – 5ml
US117.00
5ml
Brand Manufacturer: ALLERGAN INC
Please click the currency converter flag in the top corner of the screen for the exchange rate from USD-CAD.
Compare CAD-USA drug prices at GoodRx.com
This order requires you to submit a prescription. Upload here. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States. | Fulfillment policy See More.Bimatoprost (Ophthalmic), Latisse/Lumigan/Vistitan/Durysta
Description: Antiglaucoma, Prostaglandin Analogue
Brand Name US: Latisse
Canadian Equivalent: Lumigan
CSA NA – FDA Approved – REMS (N) – Can Ship
How Does It Work
Bimatoprost (Ophthalmic), Latisse/Lumigan/Vistitan/Durysta is a synthetic analog of prostaglandin with ocular hypotensive activity and decreases intraocular pressure by increasing the outflow of aqueous humor. Durysta, Latisse, Lumigan, Vistitan, Bimatoprost may increase the percent and duration of hairs in the growth phase, resulting in eyelash growth.
Indications For Use
Officially indicated for elevated intraocular pressure and hypotrichosis of the eyelashes depending on the formulation used.
Precautions
Risk of bacterial keratitis if multiple ophthalmic solutions are used
Use with caution in patients with limited corneal endothelial cell reserve, risk factors for macular edema
May permanently change the color of the iris, eyelid, periorbital tissue, and eyelashes
Usual Dosing
Elevated Intraocular Pressure
Implant (Durysta): Insert 1 implant (10 mcg) in the anterior chamber of the affected eye. Limit to a single implant per eye; do not re-administer to an eye that has received a prior implant.
Solution (Lumigan; Vistitan): Instill 1 drop into the affected eye(s) once daily in the evening; do not exceed once-daily dosing (may decrease IOP-lowering effect). If used with other topical ophthalmic agents, separate administration by at least 5 minutes.
Hypotrichosis of the Eyelashes
Solution (Latisse): Place one drop on the applicator and apply evenly along the skin of the upper eyelid at the base of eyelashes once daily at bedtime; repeat procedure for the second eye (use a clean applicator).
Pharmacist Tips On Using
When instill/inserting solutions and/or implants into the eye make sure to do so under sterile conditions. When using Durysta, insert the implant immediately after opening the foil pouch. Keep your body including your head in an upright position for ≥1 hour after administration. Make sure to remove make-up and contact lenses prior to instilling solution to the eye and/or eyelashes. When using Latisse, use the sterile applicator provided only; do not use other brushes or applicators. Use a tissue or cloth to blot any excess solution on the outside of the upper eyelid margin; do not apply to the lower eyelash line. Do not reuse applicators; use a new applicator for the second eye. Applying more than once nightly will not increase eyelash growth; eyelash growth is expected to return to baseline when therapy is discontinued. May reinsert contacts 15 minutes after application. When using Lumigan; Vistitan, wait at least 5 minutes between drops of using multiple combinations of glaucoma eye medications. Also make sure to remove your contact eye lenses prior to administration and wait 15 minutes before reinserting them.
Side Effects
The frequency of side effects is based on the use of Lumigan unless otherwise noted:
>10%:
Dermatologic: Increased growth in the number of eyelashes
Ophthalmic: Conjunctival hyperemia (25% to 45%; Latisse: <4%), eye pruritus (>10%; Latisse: <4%)
Special note for customers.
*All orders are limited to a 90 day supply. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States.Disclaimer
All information contained in and provided by ZIPRx.ca is provided for educational purposes only. This information should not be used for the diagnosis or treatment of any health problem or disease. This information is not intended to replace clinical judgement or guide individual patient care in any manner. The User is hereby notified that the information contained herein may not meet the user’s needs. The User is advised that, although the information is derived from medical research and literature, we cannot guarantee either its correctness, comprehensiveness or currency. ZIPRx nor any of its affiliated parties shall be liable for any damage resulting in whole or part from any User’s use of or reliance upon the information contained within.Shipping Details
All orders received prior to 3:00 pm PST on business days will ship the following day; peak season and holiday shipping days may require an additional 24 hours. Our Pharmacy will make every effort to ship each order as quickly as possible. Orders received over the weekend will ship the following Monday as our shipping department is closed Saturday and Sunday. We offer shipping via Canada Post within Canada. We offer shipping via USPS and UPS within the continental United States and worldwide. Expedited shipping options are available at checkout if you want to receive your order sooner.Related products
Brand Name
Allergies
Brand Name