Compare drug prices at GoodRx.ca
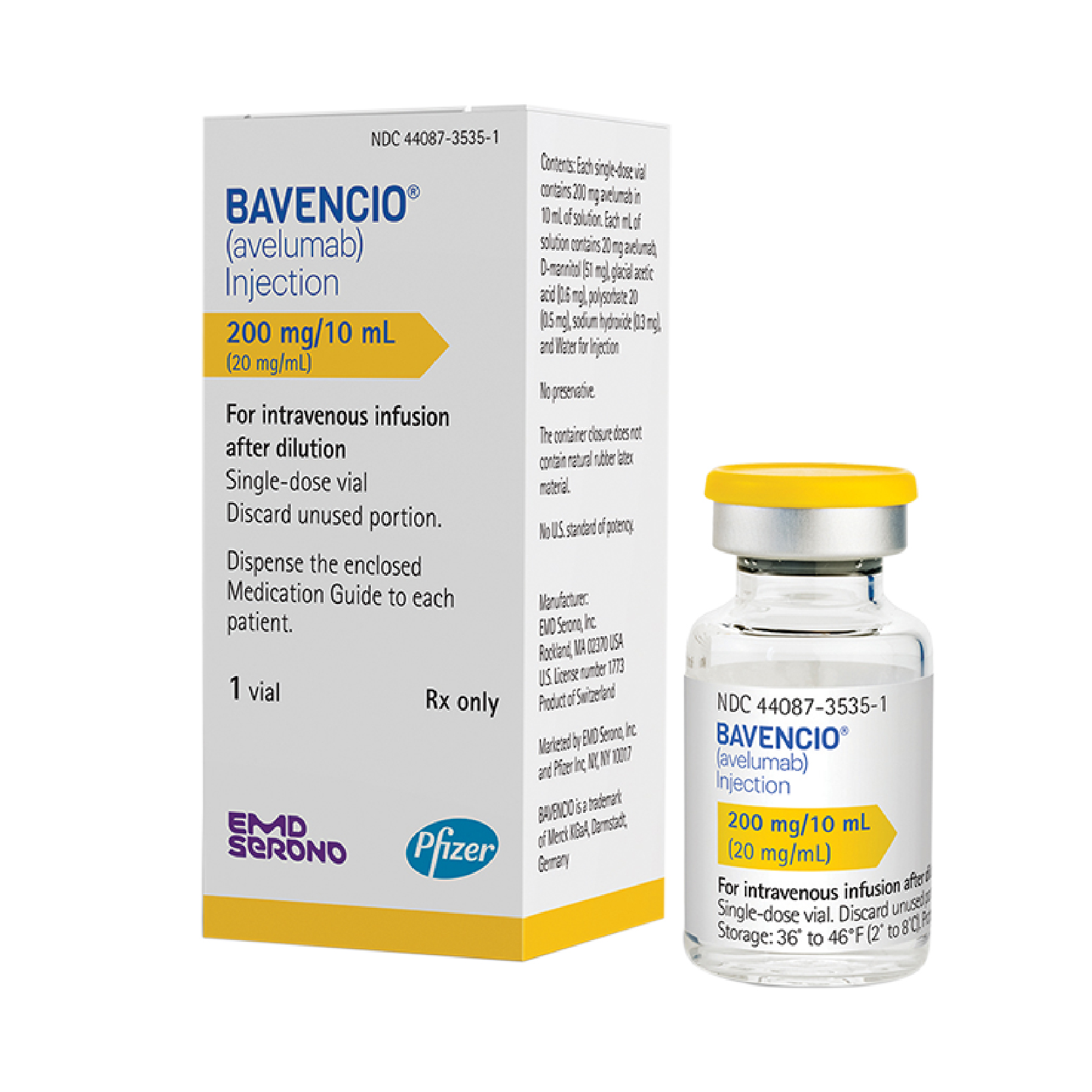
Bavencio (Avelumab) Vial 20mg/ml – 1 ea
US1,950.00
1 EA
Brand Manufacturer: EMD SERONO + PFIZER CANADA
Please click the currency converter flag in the top corner of the screen for the exchange rate from USD-CAD.
Compare CAD-USA drug prices at GoodRx.com
This order requires you to submit a prescription. Upload here. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States. | Fulfillment policy See More.Avelumab, Bavencio
Description: Antineoplastic Agent, Anti-PD-L1 Monoclonal Antibody; Immune Checkpoint Inhibitor
CSA NA – FDA Approved – REMS (N) – Can Ship
How Does It Work
Avelumab, Bavencio contains a fully human anti-programmed death-ligand (PD-L1) immunoglobulin G1 (IgG1) monoclonal antibody. The drug binds to programmed cell death 1 protein (PD-1) and controls PD-L1 interactions. It also enables the activation of T-cells and the adaptive immune system.
Indications For Use
Merkel cell carcinoma, renal cell carcinoma, urothelial carcinoma
Precautions
Before using Avelumab, Bavencio make sure your physician is aware of any allergies or medications you currently take, pre-existing medical conditions and activities you perform. Avelumab, Bavencio can remove immune response inhibition, thus potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Avelumab, Bavencio can cause myocarditis, pericarditis, and vasculitis. Avelumab, Bavencio may cause dermatological toxicity including immune-mediated rash or dermatitis. Bavenico has been associated with immune-mediated endocrinopathies including adrenal insufficiency, diabetes mellitus, hypophysitis and thyroid disorders. Avelumab, Bavencio can also cause immune-mediated colitis, pancreatitis, gastritis, and duodenitis have been reported. Immune-mediated hepatitis and nephrotoxicity have occurred with Bavenico therapy, including fatality. Immune-mediated uveitis, iritis, and other ocular inflammatory toxicities may occur with Bavenico. Some cases can be associated with retinal detachment. Pulmonary toxicity due to immune-mediated pneumonitis has been observed, including grade 2 to 4 and fatal cases.
Usual Dosing
Usual (IV): Administer 800mg IV once every 2 weeks until disease progression or unacceptable toxicity.
Pharmacist Tips On Using
How to Use: Infuse over 60 minutes through a 0.2 micron sterile, nonpyrogenic, low-protein binding inline filter. Do not infuse other medications through the same infusion line.
Side Effects
Hypertension, peripheral edema, skin rash, weight loss, abdominal pain, constipation, decreased appetite, diarrhea, increased serum lipase, nausea, vomiting, urinary tract infection, anemia, lymphocytopenia, thrombocytopenia, increased serum alanine aminotransferase (20%), increased serum aspartate aminotransferase, dizziness, fatigue, arthralgia, musculoskeletal pain, cough, dyspnea, infusion-related reaction, cellulitis, pruritus, dehydration, hyperglycemia, hyponatremia, hypothyroidism, increased amylase, colitis, intestinal obstruction, hematuria, urinary tract hemorrhage, neutropenia, increased serum bilirubin, headache
Note this is not a complete list of side effects, only the most common.
Frequently Asked Questions
What do I do if the side effects are not tolerable?
If the side effects are causing a lot of distress speak to our physician as soon as possible to discuss options.
Special note for customers.
*All orders are limited to a 90 day supply. *Prescriptions on this website are fulfilled and from Canada and depending on the medication may originate from Australia, New Zealand United Kingdom, Canada or the United States.Disclaimer
All information contained in and provided by ZIPRx.ca is provided for educational purposes only. This information should not be used for the diagnosis or treatment of any health problem or disease. This information is not intended to replace clinical judgement or guide individual patient care in any manner. The User is hereby notified that the information contained herein may not meet the user’s needs. The User is advised that, although the information is derived from medical research and literature, we cannot guarantee either its correctness, comprehensiveness or currency. ZIPRx nor any of its affiliated parties shall be liable for any damage resulting in whole or part from any User’s use of or reliance upon the information contained within.Shipping Details
All orders received prior to 3:00 pm PST on business days will ship the following day; peak season and holiday shipping days may require an additional 24 hours. Our Pharmacy will make every effort to ship each order as quickly as possible. Orders received over the weekend will ship the following Monday as our shipping department is closed Saturday and Sunday. We offer shipping via Canada Post within Canada. We offer shipping via USPS and UPS within the continental United States and worldwide. Expedited shipping options are available at checkout if you want to receive your order sooner.Related products
Brand Name